Featured
January 1, White House unveils COVID-19 vaccination plans for children ages 5 to 11
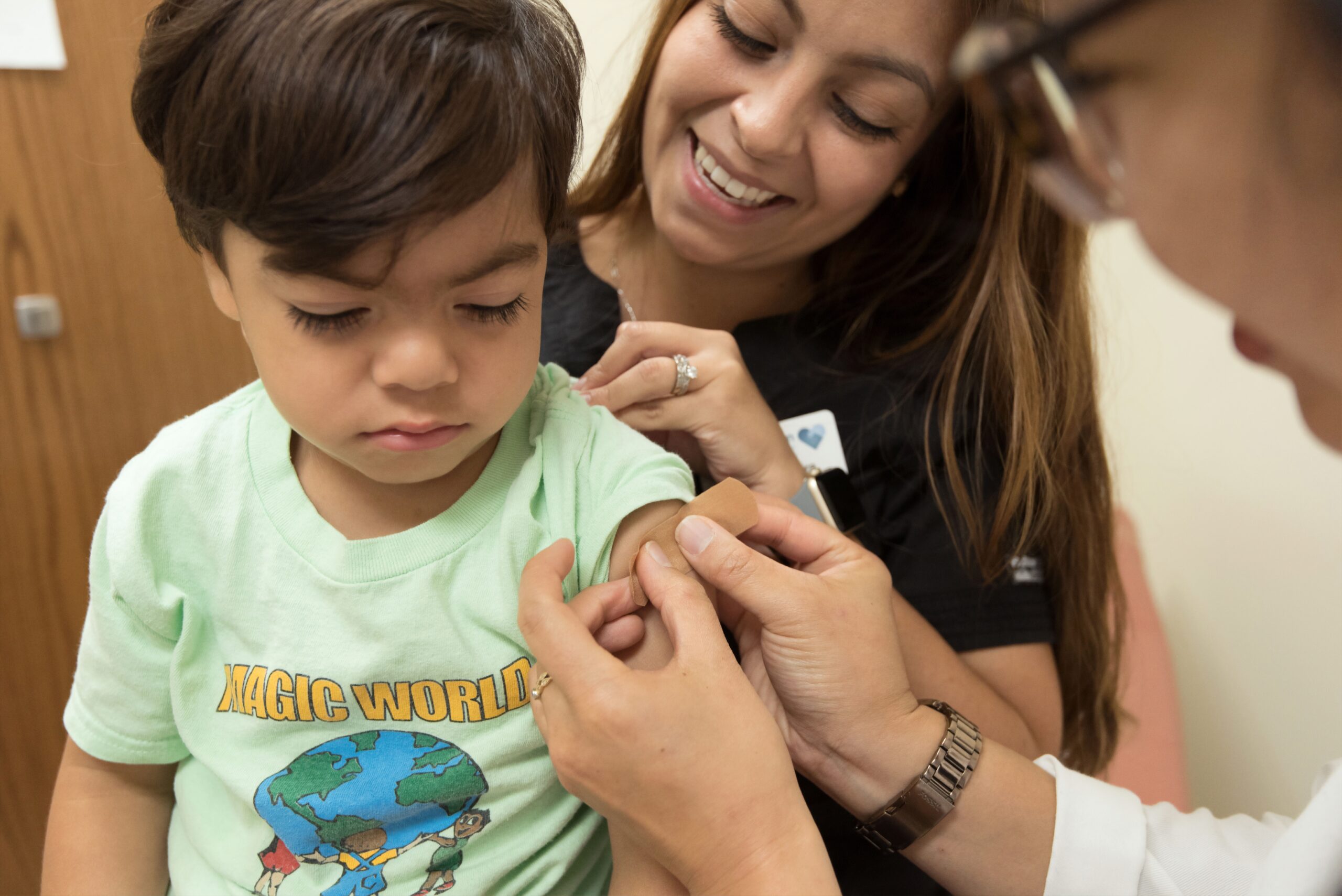
The White House revealed its plans to roll out COVID-19 vaccines for children ages 5 to 11, pending approval from the Food and Drug Administration (FDA).
“We know millions of parents have been waiting for COVID-19 vaccines for kids in this age group. And should the FDA and (US Centers for Disease Control and Prevention) authorize the vaccine, we will be ready to get shots in arms,” White House Covid-19 response director Jeff Zients said at a press briefing on Wednesday.
If the FDA authorizes the plan, the Biden administration will equip over 25,000 pediatric and primary care offices, hundreds of community and rural health centers, and thousands of pharmacies.
“Kids have different needs than adults and our operational planning is geared to meet those specific needs, including by offering vaccinations in settings that parents and kids are familiar with and trust,” Zients said.
As of Wednesday morning, 23.2% of the eligible population (about 66 million people) has not received the COVID-19 vaccination.
Source:
White House details plan to roll out Covid-19 vaccines for children ages 5 to 11

-

 Entertainment3 years ago
Entertainment3 years agoWhoopi Goldberg’s “Wildly Inappropriate” Commentary Forces “The View” into Unscheduled Commercial Break
-

 Entertainment2 years ago
Entertainment2 years ago‘He’s A Pr*ck And F*cking Hates Republicans’: Megyn Kelly Goes Off on Don Lemon
-

 Featured3 years ago
Featured3 years agoUS Advises Citizens to Leave This Country ASAP
-

 Featured3 years ago
Featured3 years agoBenghazi Hero: Hillary Clinton is “One of the Most Disgusting Humans on Earth”
-

 Entertainment2 years ago
Entertainment2 years agoComedy Mourns Legend Richard Lewis: A Heartfelt Farewell
-

 Latest News2 years ago
Latest News2 years agoNude Woman Wields Spiked Club in Daylight Venice Beach Brawl
-

 Featured3 years ago
Featured3 years agoFox News Calls Security on Donald Trump Jr. at GOP Debate [Video]
-

 Latest News2 years ago
Latest News2 years agoSupreme Court Gift: Trump’s Trial Delayed, Election Interference Allegations Linger
Jim
October 21, 2021 at 1:07 pm
How many must die so these aggressive drug pushers can become richer and more arrogant
Who me
October 27, 2021 at 3:35 pm
Just posted a comment on middlesmericanews.com. I posted and was told I already posted the same comment. What a bunch of bull. Amazing how much cheating dems and fake news will step in to silence comments they don’t like.